 All correlations deviate by less than 0.15 % from tabulated values. Measured values span several orders of magnitude. Viscosity is measured using a viscometer. For instance, honey has a much higher viscosity than water. It corresponds roughly to the intuitive notion of a fluid's 'thickness'. 4.This calculator gathers several correlations to estimate the thermodynamic and transport properties of dry air at atmospheric pressure, for the range 200 - 400 K. Dynamic viscosity is a material property which describes the resistance of a fluid to shearing flows. In Calvin, we get these equals to zero plus 2 73 0.1 is equals 2 to 73.1 Calvin. T is called 20 Now convert these equals to zero. Prandtl number density kinematic viscosity thermal diffusivity of dry air at temperatures at one atmosphere from 175 K to 1900 K. No, we will calculate the charismatic with capacity for temperature. dynamic viscosity thermal conductivity - a measure of how quickly a material can absorb heat from its surroundings. We have charismatic viscosity musicals to 1.33 into 10 to the power of minus 5 m were per second. For now, consider the table properties of air at atmospheric pressure at temperature T is equals to zero disintegrate at these equals to zero degrees Integrate. We've heard it by one plus as divided by the late this equates ambiguous a number. 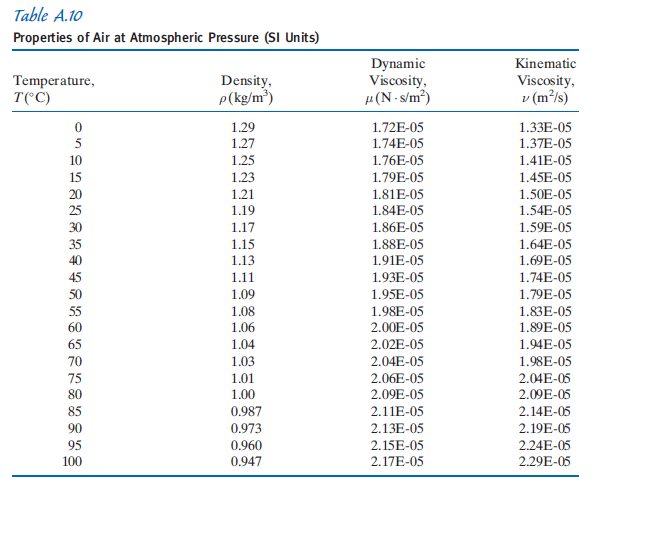
Kill him to the part of one by two Kelvin to report of one by two, therefore equation of chromatic viscosity can be written as musicals, too. So from this we get B is equal to 4.134 point 13 into 10 to the part of minus nine minus nine metre squared per second meters were per second. Multiple anyway to 87.5 divided by 101.33 into 10 to the product three. So putting the values we we get B is equal to 1.48 5458 into 10 to the part of minus six. Yeah, whole liberated way be divided Priority p divided by authority upon rearrange, invigorate new musicals too would be We are divided man be into T to the part of three Where to divided by one plus estimated t or this weekend, right? Has be t to the power of three Bed two divided by one plus s t a r p Here here b is equal to We are divided by being and it is a constant No, we need to calculate to be using the formula physicals to be at the very happy. New use equals two b t to the power one by two, divided by one plus as the very liberty. Two new musicals, too mu divided by through their music dynamic, constitute who is that density? Later, this question be it was a number three now putting the way, putting a question one and to any question, three bigot. Also, we know that the charismatic risk capacity can be written as new is equals. To be divided by RT like this is a question number two, and this is a question number one. No ideal gas equation by real gas equation Weekend right B is equal to row Artie, or role is equal. 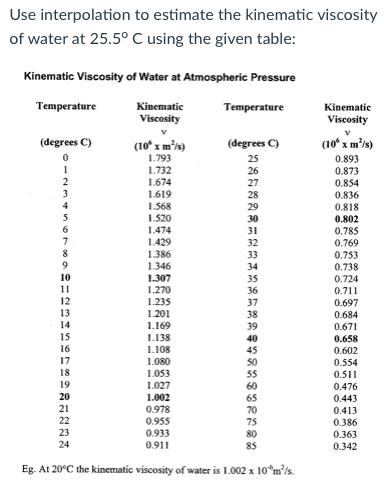 
Why wait into 10 to the part of minus six kg per meter? Second Calvin to the part of one by two and, as is equals 210.4 Kelvin. 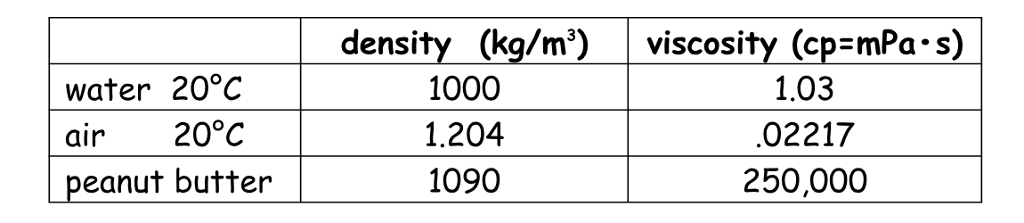
B and S are constant and T is the temperature where B can be written as 1.4. The viscosity of air depends mostly on the temperature. Muniz equals two mhm b t to the power of one way to divided by one plus as divided by t. Kinematic viscosity is the dynamic viscosity divided by the density (typical. First calculate the dynamic viscosity using the following formula, using the given values, T 0. Assuming the gas to be ideal, we can write. Water with dynamic viscosity 1 Pas Air with dynamic viscosity 0.018 Pas Mercury with dynamic viscosity 1.526 Pas. Check by using the equation to compute the kinematic viscosity of air at $0^$, using the equation and the data in Table A.10.īy Sutherland Equation. Develop an equation in SI units for kinematic viscosity versus temperature for air at atmospheric pressure. The variation with temperature of the viscosity of air is represented well by the empirical Sutherland correlationīest-fit values of $b$ and $S$ are given in Appendix A. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
